How can occur zero resistance and what is the reason of these phenomena?
Superconductivity was not sufficiently explained until 1957 when John Bardeen and his graduate assistants Leon Cooper and John Schrieffer proposed a microscopic explanation that would later be their namesake: the BCS Theory. This theoretical explanation later earned them the Nobel prize.
The BCS Theory is, in its simplest form, actually contradictory to our crude macroscopic view expressed earlier. As discussed earlier, superconductivity arises because electrons do not interact destructively with atoms in the crystal lattice of the material. The BCS Theory says that electrons do actually interact with the atoms, but constructively.
Cooper realized that atomic lattice vibrations were directly responsible for unifying the entire current. They forced the electrons to pair up into teams that could pass all of the obstacles which caused resistance in the conductor. These teams of electrons are known as Cooper pairs. Cooper and his colleagues knew that electrons which normally repel one another must feel an overwhelming attraction in superconductors. The answer to this problem was found to be in phonons, packets of sound waves present in the lattice as it vibrates. Although this lattice vibration cannot be heard, its role as a moderator is indispensable.
According to the theory, as one negatively charged electron passes by positively charged ions in the lattice of the superconductor, the lattice distorts. This in turn causes phonons to be emitted which form a trough of positive charges around the electron.
Before the electron passes by and before the lattice springs back to its normal position, a second electron is drawn into the trough. It is through this process that two electrons, which should repel one another, link up. The forces exerted by the phonons overcome the electrons' natural repulsion. The electron pairs are coherent with one another as they pass through the conductor in unison. The electrons are screened by the phonons and are separated by some distance. When one of the electrons that make up a Cooper pair and passes close to an ion in the crystal lattice, the attraction between the negative electron and the positive ion cause a vibration to pass from ion to ion until the other electron of the pair absorbs the vibration. The net effect is that the electron has emitted a phonon and the other electron has absorbed the phonon. It is this exchange that keeps the Cooper pairs together. It is important to understand, however, that the pairs are constantly breaking and reforming. Because electrons are indistinguishable particles, it is easier to think of them as permanently paired. Figure illustrates how two electrons, called Cooper pairs, become locked together.
By pairing off two by two the electrons pass through the superconductor more smoothly. The electron may be thought of as a car racing down a highway. As it speeds along, the car cleaves the air in front of it. Trailing behind the car is a vacuum, a vacancy in the atmosphere quickly filled by inrushing air. A tailgating car would be drawn along with the returning air into this vacuum. The rear car is, effectively, attracted to the one in front. As the negatively charged electrons pass through the crystal lattice of a material they draw the surrounding positive ion cores toward them. As the distorted lattice returns to its normal state another electron passing nearby will be attracted to the positive lattice in much the same way that a tailgater is drawn forward by the leading car.
The BCS theory successfully shows that electrons can be attracted to one another through interactions with the crystalline lattice. This occurs despite the fact that electrons have the same charge. When the atoms of the lattice oscillate as positive and negative regions, the electron pair is alternatively pulled together and pushed apart without a collision. The electron pairing is favorable because it has the effect of putting the material into a lower energy state. When electrons are linked together in pairs, they move through the superconductor in an orderly fashion.
As long as the superconductor is cooled to very low temperatures, the Cooper pairs stay intact, due to the reduced molecular motion. As the superconductor gains heat energy the vibrations in the lattice become more violent and break the pairs. As they break, superconductivity diminishes. This explains (roughly) why superconductivity requires low temperatures- the thermal vibration
a) Electrons carrying an electrical current through a metal wire typically encounter resistance, which is caused by collisions and scattering as the particles move through the vibrating lattice of metal atoms and electrical resistance occurs.
b) As the metal is cooled to low temperatures, the lattice vibration slows. A moving electron attracts nearby metal atoms, which create a positively charged wake behind the electron. This wake can attract another nearby electron
c) The two electrons form weak bond, called a Cooper pair, which encounters less resistance than two electrons moving separately. When more Cooper pairs form, they behave in the same way.
d) If a pair is scattered by an impurity, it will quickly get back in step with other pairs. This allows the electrons to flow un disturb through the lattice of metal atoms. With no resistance, the current may persist for years.
of the lattice must be small enough to allow the forming of Cooper pairs. In a superconductor, the current is made up of these Cooper pairs, rather than individual electrons
This BCS theory prediction of Cooper pair interaction with the crystal lattice has been verified experimentally by the isotope effect. That is, the critical temperature of a material depends on the mass of the nucleus of the atoms. If an isotope is used (neutrons are added to make it more massive), the critical temperature decreases. This effect is most evident in Type-I, and appears only weakly in Type-II.
This superconductivity of Cooper pairs is somewhat related to Bose-Einstein Condensation. The Cooper pairs act somewhat like bosons, which condense into their lowest energy level below the critical temperature, and lose electrical resistance.
The BCS Theory did exactly what a physical theory should do: it explained properties already witnessed in experiment, and it predicted experimentally verifiable phenomena. Though its specific quantitative elements were quite limited in their application (it only explained Type-I s-wave superconductivity), its essence was quite broad and has been modified applied to various other superconductors, such as Type-II perovskites.
Here the transition temperature Tc varies as where M is the mass of an isotope of a particular element. This pointed to the importance of lattice vibrations (whose frequency would be proportional to ) in mediating superconductivity.
In fact in the superconducting state the resistance falls to very small value, not zero. The resistance of any specimen may always be just less than the sensitivity of our apparatus allows us to detect. A more sensitive test, however, is to start a curret flowing round a closed superconducting ring and then see whether there is any decay in the current after a long period of time. Suppose the self inductance of the ring is L; then if at time t=0 we start a current i(0) flowing round the ring, at later time t the current will have decayed to
i(t) =i(0) e^–(R/L) t (2.2)
where R is the resistance of the ring. We can not measure the current into the circuit but can measure the magnetic field that the circulating current produces and see if the decays with time. The measurement of the magnetic field does not draw energy from the circuit, and we should be able to observe whether the current circulates indefinitely (Equation 2.2).
As can bee seen from equation for the smaller the inductance L of the circuit the more rapid the decay of current for a given value of resistance R and the more sensitive experiment
Wednesday, January 11, 2012
Superconducting Transition Temperature
The temperature at which a superconductor loss electrical resistance is called its superconducting “transition temperature” or “critical temperature” and written as Tc, is different for each metal (Table 2.1). The transition is so sudden and complete that appears to different phase of matter. Above a critical temperature Tc the properties of metal are completely normal; below Tc superconducting properties are displayed, the most dramatic of which is the absence of any measurable DC electrical resistance[23].
In general the transition temperature is not very sensitive to small amounts of impurity, but the superconductivity of a few metals, such as iridium and molybdenum, which in the pure state have very low transition temperature, may be destroyed by presence of minute quantities of magnetic impurities. Such elements, therefore, only exhibit superconductivity if they are extremely pure, and specimens of these metals of normal commercial purity are not superconductors. Not all pure metals have been found to be superconductors; for example copper, iron and sodium have not shown superconductivity down to the lowest temperature to which they have so far been cooled.
Table 2.1 Values of Tc and Hc for the superconducting elements

Superconductivity is not a rare phenomenon; about half the metallic elements are known to be superconductors and in addition a large number of alloys are superconductors. For an alloy it is possible to be a superconductor, even it is composed of two metals which are not themselves superconductors, such as Bi- Pd.
And in general some superconductor alloys have advantageous properties for applications about critical temperature. For example niobium is the metallic element with the highest transition temperature (9.3 K), but some alloys and metallic compounds remain superconducting up to even higher temperatures . For example Nb3Sn has a transition temperature of about 18 K
The eventuation of transition temperature of superconductors may be shown differences if the sample is pure or not. The transition to the superconducting state may be extremely sharp if the specimen is pure and physically perfect on cooling. For example in a good gallium specimen, the transition has been observed to occur within a temperature range 10-5 degrees. Adversely, if the specimen is impure or has a disturbed crystal structure the transition may be considerably broadened.
 Figure 2.4 -The transition temperature for pure and impure superconductor metals
Figure 2.4 -The transition temperature for pure and impure superconductor metals
In general the transition temperature is not very sensitive to small amounts of impurity, but the superconductivity of a few metals, such as iridium and molybdenum, which in the pure state have very low transition temperature, may be destroyed by presence of minute quantities of magnetic impurities. Such elements, therefore, only exhibit superconductivity if they are extremely pure, and specimens of these metals of normal commercial purity are not superconductors. Not all pure metals have been found to be superconductors; for example copper, iron and sodium have not shown superconductivity down to the lowest temperature to which they have so far been cooled.
Table 2.1 Values of Tc and Hc for the superconducting elements
Superconductivity is not a rare phenomenon; about half the metallic elements are known to be superconductors and in addition a large number of alloys are superconductors. For an alloy it is possible to be a superconductor, even it is composed of two metals which are not themselves superconductors, such as Bi- Pd.
And in general some superconductor alloys have advantageous properties for applications about critical temperature. For example niobium is the metallic element with the highest transition temperature (9.3 K), but some alloys and metallic compounds remain superconducting up to even higher temperatures . For example Nb3Sn has a transition temperature of about 18 K
The eventuation of transition temperature of superconductors may be shown differences if the sample is pure or not. The transition to the superconducting state may be extremely sharp if the specimen is pure and physically perfect on cooling. For example in a good gallium specimen, the transition has been observed to occur within a temperature range 10-5 degrees. Adversely, if the specimen is impure or has a disturbed crystal structure the transition may be considerably broadened.
 Figure 2.4 -The transition temperature for pure and impure superconductor metals
Figure 2.4 -The transition temperature for pure and impure superconductor metals
Tuesday, January 10, 2012
Electrical Resistivity
How can a metal have infinite conductivity?
In fact, the electrical resistivity of all metals and alloys decreases when they are cooled. So firstly we have to think what causes a conductor to have resistance.
A metal consists of a lattice of atoms , each with a shell of electrons. This can also be known as a positive ionic lattice. A normal metal consist of a regular crystalline lattice of positively charged ions and gas of free, on –interacting conduction electrons that fill the inter ionic space of lattice. If there is one electron per ion, we should have approximately Because of the electrons are the opposite charge from the ions, the total charge is balanced and metal is electrically neutral. When an electrical potential difference is applied across the metal, the electrons drift from one end of the conductor to the other under the influence of the electrical field. Because of the external force accelerate the free electrons a current flow occurs within the metal.
So the current in a conductor is carried by “conduction electrons” which are free to move through the material. An electron is able to pass through a perfect crystal without any loss of momentum in its original direction . However, any fault in the periodicity of the crystal will scatter the electron and introduce some resistance. So there are two effects which can spoil the perfect periodicity of crystal lattice and so introduce resistance.
1. At temperatures above absolute zero the atoms are vibrating and electrons equilibrium positions will be displaced by various amounts. As we increase the temperature, the electrons also get scattered by thermal vibrations of the lattice, called phonons, so the resistivity rises with temperature. This contribution is called phonon resistivity, . When the temperature is lowered, the thermal vibrations of atoms decreases and the conduction electrons are less frequently scattered.
A phonon, is a quantum of energy, relating to a mode of vibration occurring in a rigid crystal lattice, such as the atomic lattice of a solid
Figure 2.1- The schematic diagram of phonon resistivity
2. Foreign atoms or other defects (imperfections in periodicity like dislocations) randomly distributed can interrupt the perfect periodicity. As every real metal contains some imperfections and impurities, observes some finite resistivity at very low temperatures (extrapolated T=0) . Therefore the electrons, in addition to being scattered bye the impurities ,and this impurity scattering is more or less independent of temperature. As a result there is certain resistivity called “residual resistivity”, , which remains even at the lowest temperatures. The more impure the metal, the larger will be its residual resistivity. (Figure 2.2)
 Figure 2.2- Variation of resistance of metals with temperature
Figure 2.2- Variation of resistance of metals with temperature
Near room temperatures, the thermal motion of ions is the primary source of scattering of electrons (due to destructive interference of free electron waves on non-correlating potentials of ions), and is thus the prime cause of metal resistance. Imperfections of lattice also contribute into resistance, although their contribution in pure metals is negligible.
The larger the cross-sectional areas of the conductor, the more electrons are available to carry the current, so the lower the resistance. The longer the conductor, the more scattering events occur in each electron's path through the material, so the higher the resistance.
And, some metals ,however, show a very remarkable behavior; when they are cooled their electrical resistance decrease in the usual way, but on reaching a temperature a few degrees above absolute zero they suddenly lose all trace of electrical resistance (Figure 2.3) . They are then said to have passed into the superconducting state. And this transformation to the superconducting state may occur even if the metal is so impure that it would otherwise have had large residual resistivity.
 Figure 2.3 The difference of behavior ,non-superconductive metal and superconductor metal about loss of resistance at low temperatures.
Figure 2.3 The difference of behavior ,non-superconductive metal and superconductor metal about loss of resistance at low temperatures.
In fact, the electrical resistivity of all metals and alloys decreases when they are cooled. So firstly we have to think what causes a conductor to have resistance.
A metal consists of a lattice of atoms , each with a shell of electrons. This can also be known as a positive ionic lattice. A normal metal consist of a regular crystalline lattice of positively charged ions and gas of free, on –interacting conduction electrons that fill the inter ionic space of lattice. If there is one electron per ion, we should have approximately Because of the electrons are the opposite charge from the ions, the total charge is balanced and metal is electrically neutral. When an electrical potential difference is applied across the metal, the electrons drift from one end of the conductor to the other under the influence of the electrical field. Because of the external force accelerate the free electrons a current flow occurs within the metal.
So the current in a conductor is carried by “conduction electrons” which are free to move through the material. An electron is able to pass through a perfect crystal without any loss of momentum in its original direction . However, any fault in the periodicity of the crystal will scatter the electron and introduce some resistance. So there are two effects which can spoil the perfect periodicity of crystal lattice and so introduce resistance.
1. At temperatures above absolute zero the atoms are vibrating and electrons equilibrium positions will be displaced by various amounts. As we increase the temperature, the electrons also get scattered by thermal vibrations of the lattice, called phonons, so the resistivity rises with temperature. This contribution is called phonon resistivity, . When the temperature is lowered, the thermal vibrations of atoms decreases and the conduction electrons are less frequently scattered.
A phonon, is a quantum of energy, relating to a mode of vibration occurring in a rigid crystal lattice, such as the atomic lattice of a solid
Figure 2.1- The schematic diagram of phonon resistivity
2. Foreign atoms or other defects (imperfections in periodicity like dislocations) randomly distributed can interrupt the perfect periodicity. As every real metal contains some imperfections and impurities, observes some finite resistivity at very low temperatures (extrapolated T=0) . Therefore the electrons, in addition to being scattered bye the impurities ,and this impurity scattering is more or less independent of temperature. As a result there is certain resistivity called “residual resistivity”, , which remains even at the lowest temperatures. The more impure the metal, the larger will be its residual resistivity. (Figure 2.2)
 Figure 2.2- Variation of resistance of metals with temperature
Figure 2.2- Variation of resistance of metals with temperatureNear room temperatures, the thermal motion of ions is the primary source of scattering of electrons (due to destructive interference of free electron waves on non-correlating potentials of ions), and is thus the prime cause of metal resistance. Imperfections of lattice also contribute into resistance, although their contribution in pure metals is negligible.
The larger the cross-sectional areas of the conductor, the more electrons are available to carry the current, so the lower the resistance. The longer the conductor, the more scattering events occur in each electron's path through the material, so the higher the resistance.
And, some metals ,however, show a very remarkable behavior; when they are cooled their electrical resistance decrease in the usual way, but on reaching a temperature a few degrees above absolute zero they suddenly lose all trace of electrical resistance (Figure 2.3) . They are then said to have passed into the superconducting state. And this transformation to the superconducting state may occur even if the metal is so impure that it would otherwise have had large residual resistivity.
 Figure 2.3 The difference of behavior ,non-superconductive metal and superconductor metal about loss of resistance at low temperatures.
Figure 2.3 The difference of behavior ,non-superconductive metal and superconductor metal about loss of resistance at low temperatures.
THE PHYSICAL PROPERTIES OF SUPERCONDUCTIVITY
Provided that a material has to have the unique properties such as zero resistance to direct current; extremely high current carrying density; extremely low resistance at high frequencies; extremely low signal dispersion; high sensitivity to magnetic field; exclusion of externally applied magnetic field; rapid single flux quantum transfer; close to speed of light signal transmission, this material can be called as superconductor. Moreover, the most important of these basis properties are that; zero resistivity which means infinite conductivity under its critical temperature and the magnetic inductance which becomes zero inside the superconductor, when cooled below critical temperature in a weak external magnetic field.
The Historical Development of Superconductivity
In 1911 Kamerling Onnes from University of Leiden realized that the resistance of a thin thread of mercury sample felt to very low value when the temperature on the sample was reduced below 4.2 K (the boiling point of liquid helium). He also studied on different metals depicted in Figure1.1. As can be seen from the figure, the resistances of gold and platinum wires drop steadily to a constant value depending on the purity of the specimens. On the other hand, for mercury when the temperature is decreased, a sudden drop is observed to a low value on resistance. He named the new phenomenon as superconductivity.
Figure 1.1- The resistance versus temperature graph for some metals
After all he chose the mercury an ideal metal to explain the superconductivity phenomenon was pure enough to show obviously the steady fall of resistivity to zero. In the years to follow it was discovered that many other metallic elements exhibit superconductivity at very low temperatures .
In 1933, one of the other surprising properties of the superconductivity was discovered by Meissner and Ochsenfeld: perfect diamagnetism. According to this theory; the magnetic flux is expelled from the interior of the superconductor sample when it is cooled below the critical temperature for superconductivity in weak external magnetic fields (Figure 1.2), which is called Meissner Effect.
 Figure 1.2- Expulsion of a weak external magnetic field from the interior of the superconducting material
Figure 1.2- Expulsion of a weak external magnetic field from the interior of the superconducting material
Following the discovery of the Meissner Effect, F. and H. London proposed a simple two–fluid model in 1934. The London model was functional enough either to explain Meissner effect or predict the penetration dept , a characteristic length of penetration of the static magnetic flux into superconductor.
In 1950 Vitaly Ginzburg and Lev Landau proposed an intuitive, phenomenological theory often called a macroscopic theory ‘ Ginzburg- Landau Theory ’ of superconductivity which replied to the characteristic properties of most interesting superconducting materials including high–Tc oxides. In 1957, Alexei Abrikosov also investigated the behavior superconductors in an external magnetic field and discovered that superconductor materials can be explained in two types: type-I and type-II superconductors. While the former expels magnetic flux completely from their interior, the latter do it completely only at small fields, but partially in higher external fields. In 1957 John Bardeen, Leon Cooper and Robert Schrieffer proposed a complete microscopic theory of superconductivity that is usually referred to as the BCS Theory , of which the basis depends on the interaction of conduction electrons with elastic waves of the crystal lattice. Thus, not only does the BCS theory provide the basis for our present understanding of superconductivity in conventional materials but also some extent of the reference theory plays an important role in the on-going search for correct description of superconductivity in high–Tc cuprate oxides.
In 1962 Brian Josephson postulated fascinating quantum tunneling effects that should occur when super current tunnels through an extremely thin layer roughly 10 Å of an insulator. The prediction was confirmed within a year and the effects are known as the Josephson effects. Superconducting technology based on these effects gradually evolved and in fact Josephson junction technology represents the basis of the promising superconducting electronics nowadays.
In that year, zero resistance and perfect diamagnetism were two main problems when properties of superconductivity were tried to use in the new technology. The former needs to get very low temperature for superconducting state. In these days, although people could supply this situation with using liquid helium but that was so expensive in those years. The latter is about the condition of disruption of superconductivity for any metal, when a magnetic field applied to, or current flowed through the sample so the practical value was decreasing in applications.
There would be a lot more practical uses for superconductivity unless it were for the very high cost of liquid helium coolant. Any gas will liquefy at sufficiently low temperatures; for example, oxygen becomes liquid at 90 K and nitrogen at 77 K. Moreover, it is far less costly to fluid these gases than to liquid helium. Liquid nitrogen has a much greater cooling capacity than liquid helium. For any application in which liquid nitrogen can replace liquid helium, the refrigeration cost will be about 1000 times less .
In 1973 powerful magnets were made with using Nb-Ti which is superconductor and in fact compound had the highest critical temperature, 23 Kelvin, in all superconductors. The high- temperature history of superconductivity started in late 1986 with J. George Bednorz and Karl Muller of the IBM research laboratory in Zurich, Switzerland. They had reported the observation of superconductivity in lanthanum copper oxides doped with barium or strontium (La-Ba-Cu-O) at temperatures up to 38 K. Therefore the upper limit barrier is nearly 30 K for superconductivity that had been theoretically predicted almost 25 years earlier. This ceramic compound with perovskite structure was noted to be the first high temperature superconductor. Its multiphase nominal compound has the formula of La5xBaxCu5O5(3-y) whose critical temperature changes with regard to the change of x value. In La2-x BaxCuO4 compound the highest Tc value is obtained for x=0.15. Moreover, by using strontium instead of barium, the value of Tc can dramatically be increased up to about 36 K .
Hundreds of scientists rushed to try various chemical compounds to see which one would give the highest critical temperature. In February 1987 research groups in Alabama and Houston coordinated by K. Wu Ashburn and Paul Chu, discovered yttrium barium copper oxide ,Y-Ba-Cu-O, ceramics by using yttrium oxide instead of lanthanum oxide, with Tc=92 K . This was an important discovery owing to the existence of a superconductor with a critical temperature above that of liquid nitrogen. It is very easy to prepare Y-Ba-Cu-O ceramics by mixing calcining and oxidizing the constituent powders (Figure1.3). The multiphase nominal compounds of this sample were noted to be Y1.2Ba0.3Cu2O4-x and Y0.6Ba0.4CuO3-x. Then scientists determined that the uniphase compound called as 1: 2: 3 or Y-Ba-Cu-O is YBa2Cu3O7 by checking the ratios of barium yttrium and cooper. The difference between La-Ba-Cu-O and Y-Ba-Cu-O is pointed out to be oxygen vacancy. Depending on the vacancies, there is possibility of changing the critical temperature .
Figure1.3 - Unit cell for YBCO superconductor.
In 1987, a new chain is added to the superconductivity studies by Michael et al. who discovered the superconductivity bismuth strontium calcium copper oxide (Bi-Sr-Cu-O) ceramics. In 1988, Maeda and his group observed superconductivity at 105 K by adding calcium to this system as Bi-Sr-Ca-Cu-O . Also Tokano and his group found a stable superconductor at 110 K by adding lead to the compound . In 1992 Quidwai and his group carried out the transition at 130-140 K by adding antimony to the system .
In 1988, Sheng and Parking observed superconductivity in thallium barium calcium copper oxide (Tl-Ba-Ca-Cu-O) compound at 110 K and 125 K. Nevertheless this compound either has carcinogenic effect or was unstable thus has to be produced each time. During these passing years, studies on different samples went on. In 1993 Schilling and his group observed superconductivity in mercury barium calcium copper oxide HgBa2CaCu3O1+x compound at 133 K; similarly, Chu and his group observed superconductivity in high pressure with Hg based compounds at 150 K .
In 2001, J. Nagamatsu, J. Akimitsu and their group observed superconductivity in MgB2 compound at 39 K, which was a very important discovery because of the fact that this critical temperature was quite high for Type-I superconductors and elastic material so it had the advantage of being easy to be adapted to technology.
Table 1.1 High Critical Temperature Superconducting Compounds

In 2008, the discovery of a new family of high critical temperature iron and arsenic superconductors (AsFe) marked a second major revolution in the world of superconductivity. The new compounds, which do not contain copper (Cu) but which have oxygen (O), flour (F) or arsenic (As) and iron (Fe), will help scientists to solve some of the mysteries in the area of solid state physics and to answer some questions such as “Is the AsFe superconductor family really so different from second type superconductor?”. This point is fundamental to define a unified approach to the two families of superconducting materials.
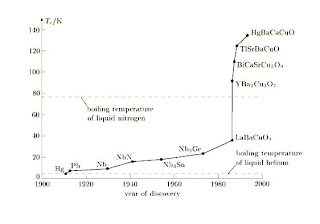 Figure 1.4- The critical temperature Tc of various superconductors plotted against their discovery date.
Figure 1.4- The critical temperature Tc of various superconductors plotted against their discovery date.
In 1933, one of the other surprising properties of the superconductivity was discovered by Meissner and Ochsenfeld: perfect diamagnetism. According to this theory; the magnetic flux is expelled from the interior of the superconductor sample when it is cooled below the critical temperature for superconductivity in weak external magnetic fields (Figure 1.2), which is called Meissner Effect.
 Figure 1.2- Expulsion of a weak external magnetic field from the interior of the superconducting material
Figure 1.2- Expulsion of a weak external magnetic field from the interior of the superconducting materialFollowing the discovery of the Meissner Effect, F. and H. London proposed a simple two–fluid model in 1934. The London model was functional enough either to explain Meissner effect or predict the penetration dept , a characteristic length of penetration of the static magnetic flux into superconductor.
In 1950 Vitaly Ginzburg and Lev Landau proposed an intuitive, phenomenological theory often called a macroscopic theory ‘ Ginzburg- Landau Theory ’ of superconductivity which replied to the characteristic properties of most interesting superconducting materials including high–Tc oxides. In 1957, Alexei Abrikosov also investigated the behavior superconductors in an external magnetic field and discovered that superconductor materials can be explained in two types: type-I and type-II superconductors. While the former expels magnetic flux completely from their interior, the latter do it completely only at small fields, but partially in higher external fields. In 1957 John Bardeen, Leon Cooper and Robert Schrieffer proposed a complete microscopic theory of superconductivity that is usually referred to as the BCS Theory , of which the basis depends on the interaction of conduction electrons with elastic waves of the crystal lattice. Thus, not only does the BCS theory provide the basis for our present understanding of superconductivity in conventional materials but also some extent of the reference theory plays an important role in the on-going search for correct description of superconductivity in high–Tc cuprate oxides.
In 1962 Brian Josephson postulated fascinating quantum tunneling effects that should occur when super current tunnels through an extremely thin layer roughly 10 Å of an insulator. The prediction was confirmed within a year and the effects are known as the Josephson effects. Superconducting technology based on these effects gradually evolved and in fact Josephson junction technology represents the basis of the promising superconducting electronics nowadays.
In that year, zero resistance and perfect diamagnetism were two main problems when properties of superconductivity were tried to use in the new technology. The former needs to get very low temperature for superconducting state. In these days, although people could supply this situation with using liquid helium but that was so expensive in those years. The latter is about the condition of disruption of superconductivity for any metal, when a magnetic field applied to, or current flowed through the sample so the practical value was decreasing in applications.
There would be a lot more practical uses for superconductivity unless it were for the very high cost of liquid helium coolant. Any gas will liquefy at sufficiently low temperatures; for example, oxygen becomes liquid at 90 K and nitrogen at 77 K. Moreover, it is far less costly to fluid these gases than to liquid helium. Liquid nitrogen has a much greater cooling capacity than liquid helium. For any application in which liquid nitrogen can replace liquid helium, the refrigeration cost will be about 1000 times less .
In 1973 powerful magnets were made with using Nb-Ti which is superconductor and in fact compound had the highest critical temperature, 23 Kelvin, in all superconductors. The high- temperature history of superconductivity started in late 1986 with J. George Bednorz and Karl Muller of the IBM research laboratory in Zurich, Switzerland. They had reported the observation of superconductivity in lanthanum copper oxides doped with barium or strontium (La-Ba-Cu-O) at temperatures up to 38 K. Therefore the upper limit barrier is nearly 30 K for superconductivity that had been theoretically predicted almost 25 years earlier. This ceramic compound with perovskite structure was noted to be the first high temperature superconductor. Its multiphase nominal compound has the formula of La5xBaxCu5O5(3-y) whose critical temperature changes with regard to the change of x value. In La2-x BaxCuO4 compound the highest Tc value is obtained for x=0.15. Moreover, by using strontium instead of barium, the value of Tc can dramatically be increased up to about 36 K .
Hundreds of scientists rushed to try various chemical compounds to see which one would give the highest critical temperature. In February 1987 research groups in Alabama and Houston coordinated by K. Wu Ashburn and Paul Chu, discovered yttrium barium copper oxide ,Y-Ba-Cu-O, ceramics by using yttrium oxide instead of lanthanum oxide, with Tc=92 K . This was an important discovery owing to the existence of a superconductor with a critical temperature above that of liquid nitrogen. It is very easy to prepare Y-Ba-Cu-O ceramics by mixing calcining and oxidizing the constituent powders (Figure1.3). The multiphase nominal compounds of this sample were noted to be Y1.2Ba0.3Cu2O4-x and Y0.6Ba0.4CuO3-x. Then scientists determined that the uniphase compound called as 1: 2: 3 or Y-Ba-Cu-O is YBa2Cu3O7 by checking the ratios of barium yttrium and cooper. The difference between La-Ba-Cu-O and Y-Ba-Cu-O is pointed out to be oxygen vacancy. Depending on the vacancies, there is possibility of changing the critical temperature .
Figure1.3 - Unit cell for YBCO superconductor.
In 1987, a new chain is added to the superconductivity studies by Michael et al. who discovered the superconductivity bismuth strontium calcium copper oxide (Bi-Sr-Cu-O) ceramics. In 1988, Maeda and his group observed superconductivity at 105 K by adding calcium to this system as Bi-Sr-Ca-Cu-O . Also Tokano and his group found a stable superconductor at 110 K by adding lead to the compound . In 1992 Quidwai and his group carried out the transition at 130-140 K by adding antimony to the system .
In 1988, Sheng and Parking observed superconductivity in thallium barium calcium copper oxide (Tl-Ba-Ca-Cu-O) compound at 110 K and 125 K. Nevertheless this compound either has carcinogenic effect or was unstable thus has to be produced each time. During these passing years, studies on different samples went on. In 1993 Schilling and his group observed superconductivity in mercury barium calcium copper oxide HgBa2CaCu3O1+x compound at 133 K; similarly, Chu and his group observed superconductivity in high pressure with Hg based compounds at 150 K .
In 2001, J. Nagamatsu, J. Akimitsu and their group observed superconductivity in MgB2 compound at 39 K, which was a very important discovery because of the fact that this critical temperature was quite high for Type-I superconductors and elastic material so it had the advantage of being easy to be adapted to technology.
Table 1.1 High Critical Temperature Superconducting Compounds

In 2008, the discovery of a new family of high critical temperature iron and arsenic superconductors (AsFe) marked a second major revolution in the world of superconductivity. The new compounds, which do not contain copper (Cu) but which have oxygen (O), flour (F) or arsenic (As) and iron (Fe), will help scientists to solve some of the mysteries in the area of solid state physics and to answer some questions such as “Is the AsFe superconductor family really so different from second type superconductor?”. This point is fundamental to define a unified approach to the two families of superconducting materials.
 Figure 1.4- The critical temperature Tc of various superconductors plotted against their discovery date.
Figure 1.4- The critical temperature Tc of various superconductors plotted against their discovery date.
Types I and II Superconductors

There are thirty pure metals which exhibit zero resistivity at low temperatures and have the property of excluding magnetic fields from the interior of the superconductor (Meissner effect). They are called Type I superconductors. The superconductivity exists only below their critical temperatures and below a critical magnetic field strength. Type I superconductors are well described by the BCS theory.
Starting in 1930 with lead-bismuth alloys, a number of alloys were found which exhibited superconductivity; they are called Type II superconductors. They were found to have much higher critical fields and therefore could carry much higher current densities while remaining in the superconducting state.
The variations on barium-copper-oxide ceramics which achieved the superconducting state at much higher temperatures are often just referred to as high temperature superconductors and form a class of their own.
Type I Superconductors
The thirty pure metals listed at right below are called Type I superconductors. The identifying characteristics are zero electrical resistivity below a critical temperature, zero internal magnetic field (Meissner effect), and a critical magnetic field above which superconductivity ceases.
The superconductivity in Type I superconductors is modeled well by the BCS theory which relies upon electron pairs coupled by lattice vibration interactions. Remarkably, the best conductors at room temperature (gold, silver, and copper) do not become superconducting at all. They have the smallest lattice vibrations, so their behavior correlates well with the BCS Theory.
While instructive for understanding superconductivity, the Type I superconductors have been of limited practical usefulness because the critical magnetic fields are so small and the superconducting state disappears suddenly at that temperature. Type I superconductors are sometimes called "soft" superconductors while the Type II are "hard", maintaining the superconducting state to higher temperatures and magnetic fields.
Type II Superconductors
 Superconductors made from alloys are called Type II superconductors. Besides being mechanically harder than Type I superconductors, they exhibit much higher critical magnetic fields. Type II superconductors such as niobium-titanium (NbTi) are used in the construction of high field superconducting magnets.
Superconductors made from alloys are called Type II superconductors. Besides being mechanically harder than Type I superconductors, they exhibit much higher critical magnetic fields. Type II superconductors such as niobium-titanium (NbTi) are used in the construction of high field superconducting magnets.Type-II superconductors usually exist in a mixed state of normal and superconducting regions. This is sometimes called a vortex state, because vortices of superconducting currents surround filaments or cores of normal material.
Subscribe to:
Posts (Atom)


